Structure of Atom
9th chemistry chapter 2 solved exercise short questions
Q#1 : What is nature of charge on cathode rays ?
Cathode rays have negative charge on them.

Q#2 : Give five characteristics of cathode rays ?
Characteristics of Cathode rays :
- Cathode rays travel in a straight line perpendicular to the cathode surface .
- They raise the temperature of the body on which they fall.
- Light is produced when these rays hit the walls of discharge tube .
- They can cast a sharp shadow .
- The nature of rays do not depend upon the nature of gas used in discharge tube .
Q#3 : The atomic symbol of a phosphorus ion is given as :
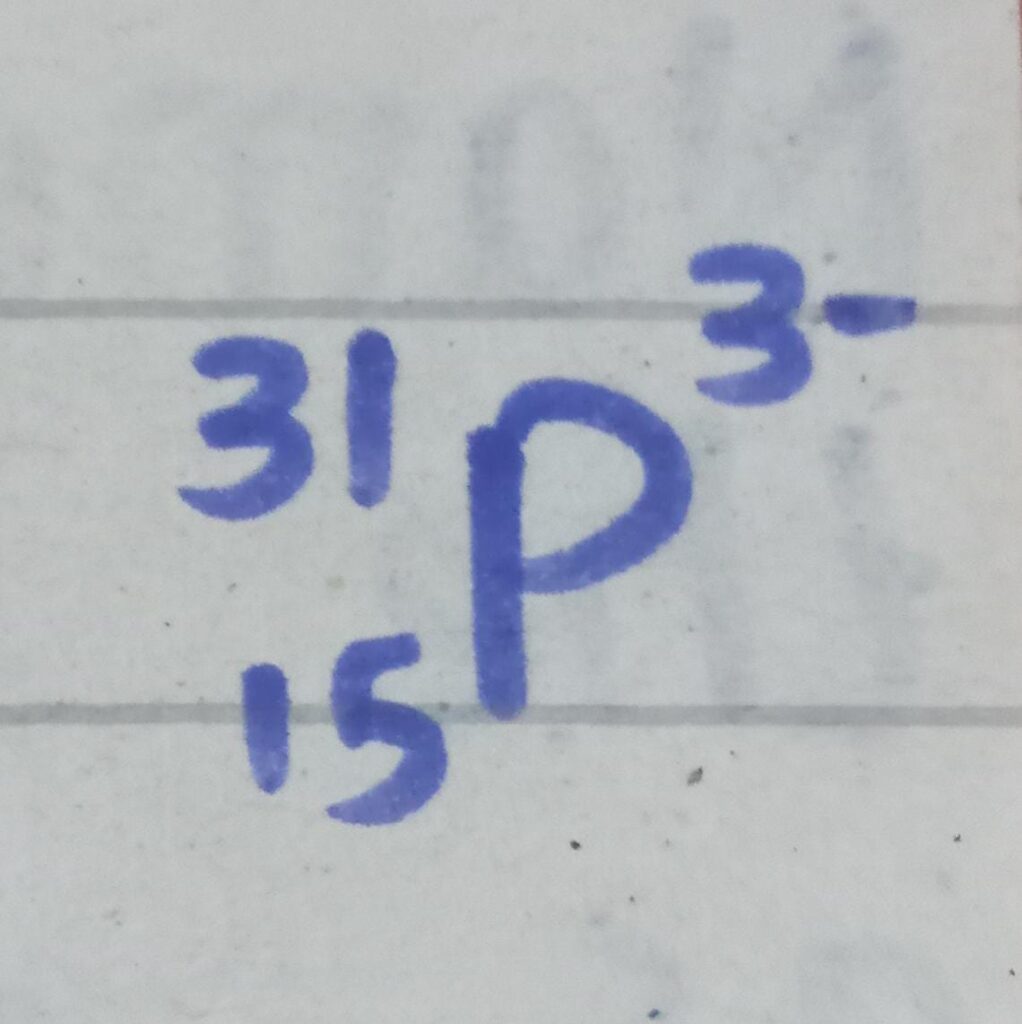
(a) : How many protons , electrons and neutrons are there in the ion ?
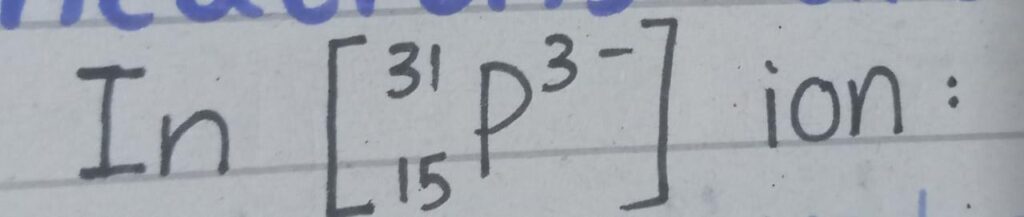
Atomic number is 15
- Number of protons = 15
It has -3 charge which shows gain of 3 electrons.
- Number of electrons = 15 + 3 = 18
- Number of neutrons = 31 – 15 = 16
(b) : What is name of the ion ?
P-3 is called phosphide ion .
(c) : Draw the electronic configuration of the ion .
The electronic configuration of P3- :
| K | L | M |
| 2 | 8 | 8 |
1s² , 2s² , 2p⁶ , 3s² , 3p⁶
(d) : Name the noble gas which has the same electronic configuration as the phosphorus ion has .
Argon (Ar)
Q#4 : Differentiate between shell and subshell with examples of each .

Q#5 : An element has an atomic number 17 . How many electrons are present in K, L and M shells of the atom ?
Atomic number = Z = 17
K = 2 electrons
L = 8 electrons
M = 7 electrons
Q#6 : Write down the electronic configuration of Al+3 . How many electrons are present in its outermost shell ?
Aluminum has 13 electrons ( atomic number = 13 ) while Al+3 ion has 10 electrons due to loss of 3 electrons.
Electronic configuration (Al+3) in shells :
| K | L |
| 2 | 8 |
Electronic configuration of Al+3 in sub-shells:
1s² , 2s² , 2p⁶
Q#7 : Magnesium has electronic configuration 2, 8, 2.
(a) : How many electrons are in outermost shell ?
The outermost shell of Mg has only two electrons.
(b) : In which subshell of the outermost shell electrons are present ?
The outermost electrons of “Mg” are present in “s” subshell of the 3rd shell “M” (3s²) because its electronic configuration is :
1s² , 2s² , 2p⁶ , 3s²
(c) : Why magnesium tends to lose electrons ?
Magnesium is an electropositive metal. It can easily lose its two outermost electrons and get charge (2+).
Q#8 : What will be the nature of charge on an atom when it loses an electron or when it gains an electron ?
- When an atom loses its electron, it becomes positively charged called Cation .
- When an atom gains electrons, it becomes negatively charged ion called Anion .
Q#9 : For what purpose is U-235 used ?
Use of U-235 :
U-235 is used to get large amount of energy which can be used to drive the turbines to generate electricity .
Q#10 : A patient has goiter . How will it be detected ?
Detection of Goiter :
I-131 is given to the patient . The amount of iodine absorbed and the radiations emitted from thyroid glands is measured to detect tumor .
Q#11: Give three properties of positive rays .
Properties of Positive Rays :
Positive rays are also called “canal rays ” .
- These rays travel in a straight line in a direction opposite to the cathode rays .
- These are positively charged rays .
- The nature of these rays depend upon the nature of gas filled in discharge tube .
Q#12 : What are the defects of Rutherford’s atomic model ?
Defects of Rutherford’s Atomic Model :
- According to this atomic model , a revolving electron being a charged particle must radiate energy continuously .By losing energy , it will ultimately fall in the nucleus which is impossible .
- Rutherford’s model of atom also suggests that there would be a continuous atomic spectrum but continuous atomic spectrum is obtained .
Q#13 : As long as electrons remains in an orbit , it does not emit or absorb energy. When does it emit or absorb energy ?
- Electrons emits energy (ΔE=E2-E1) when it jumps from higher energy level to lower energy level .
- Electron absorbs energy (ΔE=E2-E1) when it jumps from lower energy level to higher energy level .
9th chemistry chapter 2 solved exercise short questions
